核医学市場 概要
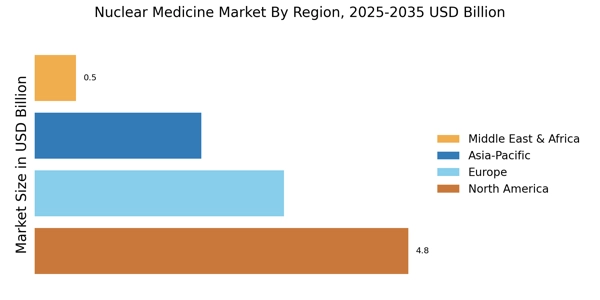
市場調査未来の分析によると、核医学市場の規模は2024年に106.3億米ドルと推定されています。核医学業界は、2025年に118.4億米ドルから2035年までに346.9億米ドルに成長すると予測されており、2025年から2035年の予測期間中に年平均成長率(CAGR)は11.3%です。北米は市場をリードしており、45%以上のシェアを持ち、約48億米ドルの収益を生み出しています。
がんの有病率の上昇、診断画像の採用の拡大、標的放射性医薬品の使用の増加は、核医学市場の主要な成長ドライバーです。早期の病気検出と精密腫瘍学への需要は、世界的に臨床採用を大幅に加速させています。
WHO(IARC)によると、2022年には世界中で約2000万件の新しいがん症例が発生し、約970万人が死亡しており、PETおよびSPECT画像への強い需要を生み出しています。IHMEの世界的な疾病負担の推定は、がん関連の死亡率が世界中で着実に上昇していることを示しており、早期診断と治療モニタリングにおける核医学の採用を強化しています。
主要な市場動向とハイライト
核医学市場は、技術の進歩と個別化医療の需要の高まりにより、 substantial growthが期待されています。
- テクネチウム-99mは、55%のシェアで放射性同位体の使用を支配し、医療システム全体で広範な診断画像手順をサポートしています。
- PET画像は、58%のシェアで手順セグメントをリードしており、優れた代謝可視化と腫瘍学アプリケーションにより推進されています。
- 北米は、先進的な医療インフラと癌の有病率に支えられ、グローバル市場で45%以上の収益シェアを持っています。
- ヨーロッパ市場は105億ユーロと評価され、強力な規制と高齢化人口の支援により30%のシェアを保持しています。
市場規模と予測
| 2024年の市場規模 | 10.63(米ドル十億) |
| 2035年の市場規模 | 34.69(米ドル十億) |
| CAGR(2025 - 2035) | 11.35% |
主要なプレーヤー
GEヘルスケア(米国)、シーメンスヘルスケア(ドイツ)、フィリップスヘルスケア(オランダ)、キヤノンメディカルシステムズ(日本)、エレクタ(スウェーデン)、バイエルAG(ドイツ)、ブラッコイメージング(イタリア)、ランセウスメディカルイメージング(米国)、ノーススター医療ラジオアイソトープ(米国)などの企業は、世界市場の主要な参加者の一部です。