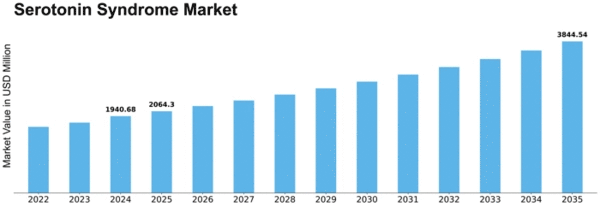
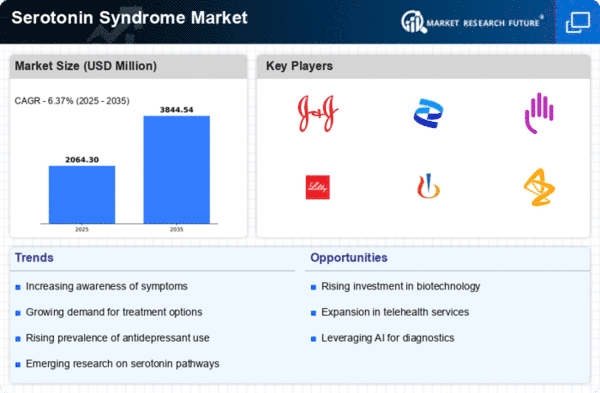
Serotonin Syndrome Size
Serotonin Syndrome Market Growth Projections and Opportunities
Serotonin syndrome is a potentially deadly disorder caused by excessive amounts of serotonin in the body, often as a result of certain drugs. Many factors influence its prevalence and management which reflects the complex relationship between pharmaceuticals, healthcare practices and patient outcomes.
The extensive use of medications affecting serotonin levels is one major market factor. For various mental health conditions, SSRIs (Selective Serotonin Reuptake Inhibitors) or SNRIs (Serotonin-Norepinephrine Reuptake Inhibitors) are commonly prescribed antidepressants. Some other drugs like some pain relievers and migraine treatments may also have an impact on serotonin levels. The increasing presence of these medications on the market raises the likelihood for cases of serotonin syndrome.
Another industry factor regarding serotonin syndrome is polypharmacy in healthcare where multiple drugs are taken at once. Different medical professionals frequently prescribe medicines to patients thus exposing them to possible drug interactions that can increase serotonin levels to dangerous limits. This problem is worsened by the absence of a centralised method of tracking prescriptions given to patients as well as their medication history making it hard to identify and mitigate SSRI risks.
Besides, there are different knowledge levels among healthcare practitioners about the illness in this market. Symptoms of this disease may be nonspecific making it difficult for accurate diagnosis because they resemble those for other medical conditions. Poor awareness and education by healthcare providers can cause delayed recognition leading to severe outcomes among victims for example implying that there needs more education within the health care sector regarding early detection and appropriate management strategies for SSRI.
Additionally, the pharmaceutical industry contributes significantly towards the market for Serotonin Syndrome. Manufacturers must ensure they provide detailed information about potential risks involved with their products relating with Serotonin Syndrome. Due to competition and profit maximization drivers, focus could inadvertently shift away from communicating these hazards well enough both to doctors as well as patients. Thus safety information should be prioritized by pharmaceutical firms collaborating with regulatory bodies and other industry stakeholders, while also providing enough warning of possible serotonin syndrome.
Further, the market is shaped by changing landscape of mental health care. The numbers of persons seeking help for mental illnesses are increasing bringing about greater demand for antidepression drugs as well as other serotonergic medications. This upward trend in prescription rates further underscores the importance of monitoring and managing the risk of serotonin syndrome in the broader healthcare market.”

















Leave a Comment