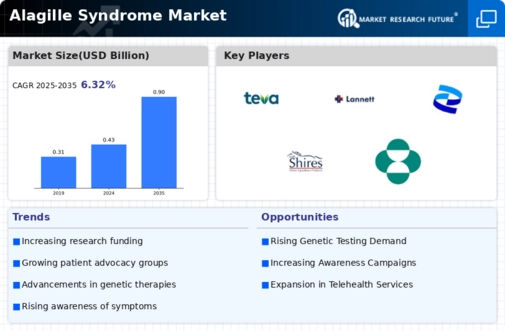
Market Share
Alagille Syndrome Market Share Analysis
The market dynamics of Alagille Syndrome unfold within the landscape of rare genetic disorders, emphasizing the challenges and opportunities associated with the diagnosis, treatment, and management of this condition. Alagille Syndrome is a rare genetic disorder characterized by liver, heart, and other organ abnormalities, affecting approximately 1 in 30,000 to 1 in 50,000 individuals. One key driver of the market is the increasing awareness and understanding of Alagille Syndrome among healthcare professionals, patients, and their families. As knowledge about this rare condition expands, the demand for effective diagnostic tools, therapeutic interventions, and supportive care options rises, shaping the market dynamics significantly.
Technological advancements in genetic testing and diagnostic imaging contribute significantly to the market dynamics of the Alagille Syndrome market. Innovations in genetic sequencing technologies allow for more accurate and comprehensive identification of mutations associated with Alagille Syndrome, facilitating early and precise diagnosis. Additionally, advanced imaging techniques, such as magnetic resonance imaging (MRI) and ultrasound, assist healthcare professionals in assessing the extent of organ involvement and guiding treatment strategies. The continuous evolution of diagnostic tools underscores the commitment of the healthcare industry to improve early detection and intervention for individuals affected by Alagille Syndrome.
Collaborations and partnerships between research institutions, healthcare organizations, and pharmaceutical companies contribute to the dynamic landscape of the Alagille Syndrome market. These alliances foster knowledge exchange, facilitate clinical trials, and drive research initiatives aimed at developing innovative therapeutic interventions. Given the rarity and complexity of Alagille Syndrome, collaborative efforts within the scientific community are instrumental in advancing our understanding of the condition, refining diagnostic approaches, and exploring potential treatment options. The synergy between different stakeholders accelerates progress in addressing the unique challenges posed by this rare genetic disorder.
The pharmaceutical industry's engagement in the development of targeted therapies for Alagille Syndrome is a significant factor shaping market dynamics. As the molecular mechanisms underlying the disorder become clearer, pharmaceutical companies invest in research and development to create therapeutic interventions that address the specific genetic defects and associated symptoms. The pursuit of novel drugs and treatment options reflects a commitment to improving the quality of life for individuals with Alagille Syndrome, driving innovation in the field and contributing to the overall growth and competitiveness of the market.
Regulatory frameworks and standards also play a critical role in guiding the market dynamics of the Alagille Syndrome market. Adherence to regulatory requirements ensures the safety and efficacy of diagnostic tests and therapeutic interventions. Regulatory oversight provides a framework for the development and approval of new drugs, diagnostic devices, and treatment protocols, ensuring that they meet rigorous standards and contribute to improved patient outcomes.
Economic factors, including healthcare spending and reimbursement policies, contribute to the market dynamics of Alagille Syndrome. The economic feasibility of diagnostic tests, medications, and treatment options influences their accessibility and adoption within healthcare systems. As stakeholders seek cost-effective solutions that align with budgetary constraints, the market responds by striving to deliver value-based care for individuals affected by Alagille Syndrome.

















Leave a Comment