Market Share
Proteinuria Treatment Market Share Analysis
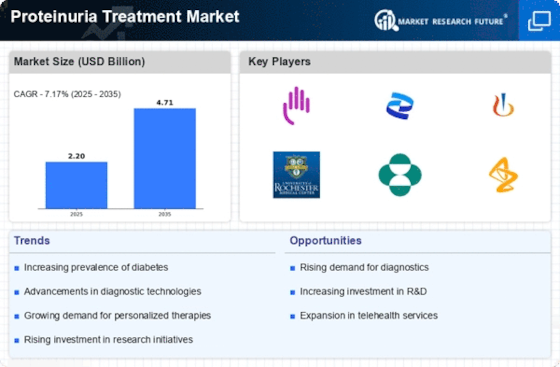
The proteinuria treatment market is witnessing significant developments driven by the increasing prevalence of kidney disorders, advances in treatment options, and focus on early intervention to control proteinuria, which is identified as a condition associated with abnormally large amounts of proteins in urine. According to recent studies, there is a major trend towards targeted therapies aimed at managing proteinuria linked to different renal conditions, such as diabetic nephropathy and glomerulonephritis, among others, along with other kidney diseases. Furthermore, the Proteinuria Treatment Market responds effectively when integrated with precision medicine approaches. Specific biomarker identification and genetic profiling enable individualized and more focused treatment approaches for those people living with proteinuria. The present market is witnessing an upsurge in the search for new therapeutic techniques beyond traditional pharmacological interventions. Lifestyle changes, dietary manipulations, and alternative strategies, including herbal supplements and nutraceuticals, are emerging as potential complementary options to pharmacotherapy in handling proteinuria. Collaborations between drug companies, research centers, and healthcare institutions are instrumental in shaping the Proteinuria Treatment Market. They encourage scientific research activities, clinical trials, and the development of innovative therapies. Furthermore, these alliances also enhance the widespread transmission of knowledge, drawing treatment guidelines and translating scientific findings into pragmatic, useful therapies. An increased understanding of the molecular as well as genetic basis for proteinuria has led to the adoption of gene/cell therapy within the proteinuria treatment market. This future direction would aim at targeting specific genetic mutations or cellular abnormalities that lead to proteinuria, thus transforming how certain genetic kidney diseases are dealt with medically. The market is responding to the growing prevalence of chronic kidney disease (CKD), which has seen an increase in cases of proteinuria. The market is focused on early detection and intervention strategies because proteinuria can be used as a marker for earlier kidney dysfunction. Some screening programs, routine urine tests, and point-of-care diagnostics help identify early signs of proteinuria, which allows timely intervention and management before the progression of kidney diseases.


















Leave a Comment