Increasing Incidence of Cancer
The rising incidence of various cancers globally appears to be a primary driver for the Global Paraneoplastic Syndrome Market Industry. As cancer cases increase, the associated paraneoplastic syndromes are also likely to rise, leading to a greater demand for diagnostic and therapeutic solutions. For instance, the World Health Organization indicates that cancer cases are projected to reach 29.5 million by 2040. This growing patient population necessitates enhanced awareness and treatment options for paraneoplastic syndromes, which could contribute to the market's expansion, with an estimated value of 460 USD Million in 2024.
Development of Targeted Therapies
The emergence of targeted therapies for cancer treatment is likely to have a profound impact on the Global Paraneoplastic Syndrome Market Industry. These therapies, designed to specifically target cancer cells, may also influence the management of paraneoplastic syndromes. For instance, monoclonal antibodies and small molecule inhibitors are being explored for their potential to alleviate symptoms associated with these syndromes. As research continues to evolve, the availability of such therapies could enhance treatment options for patients suffering from paraneoplastic syndromes, thereby driving market growth and fostering innovation in this field.
Advancements in Diagnostic Techniques
Technological advancements in diagnostic methodologies are expected to significantly influence the Global Paraneoplastic Syndrome Market Industry. Innovations such as liquid biopsies and advanced imaging techniques facilitate earlier and more accurate detection of paraneoplastic syndromes. These advancements not only improve patient outcomes but also enhance the overall efficiency of healthcare systems. For example, the integration of artificial intelligence in imaging has shown promise in identifying subtle changes associated with paraneoplastic syndromes. As these technologies become more widely adopted, the market is likely to experience growth, potentially reaching 609.8 USD Million by 2035.
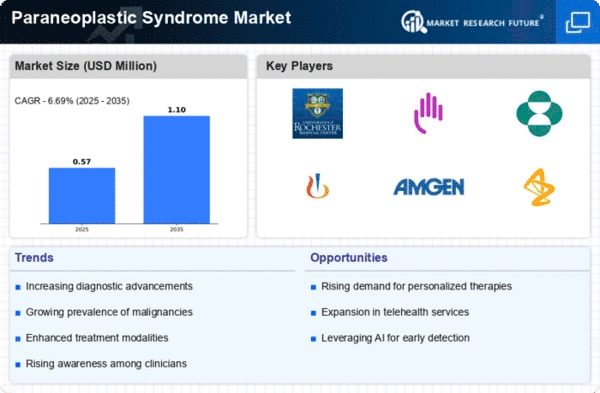
Chart Representation of Market Trends
This section includes charts that illustrate the projected growth and trends within the Global Paraneoplastic Syndrome Market Industry. The charts depict the anticipated market value increase from 460 USD Million in 2024 to 609.8 USD Million by 2035, alongside the expected CAGR of 2.6% from 2025 to 2035. These visual representations provide a clear overview of the market dynamics and growth potential, aiding stakeholders in understanding the evolving landscape of paraneoplastic syndrome management.
Rising Awareness Among Healthcare Professionals
There appears to be a growing awareness among healthcare professionals regarding paraneoplastic syndromes, which could drive the Global Paraneoplastic Syndrome Market Industry. Increased education and training initiatives are being implemented to help clinicians recognize these syndromes, leading to timely diagnosis and management. This heightened awareness may result in improved patient care and outcomes, thereby increasing the demand for related treatments and diagnostic tools. As healthcare providers become more knowledgeable about the implications of paraneoplastic syndromes, the market is likely to benefit from this trend, contributing to a projected CAGR of 2.6% from 2025 to 2035.



















Leave a Comment