Market Share
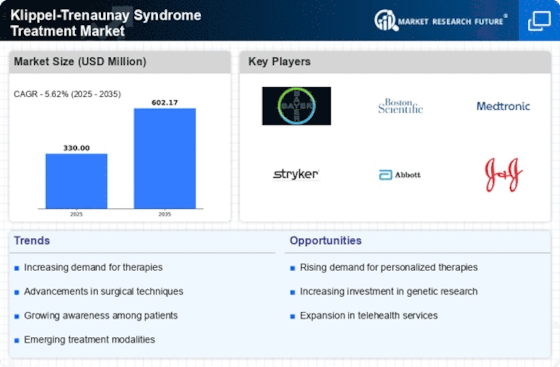
Klippel Trenaunay Syndrome Treatment Market Share Analysis
Klippel Trenaunay Syndrome (KTS) Treatment Market is evolving at a rapid pace with several companies positioning themselves strategically to gain competitive advantage and increase their market shares. At the core of these strategies are differentiated product offerings, which include developing unique treatments or therapies for Klippel Trenaunay Syndrome. Through this, specialized solutions that focus on symptom relieve, quality of life enhancements or any other aspect specific to KTS patients can be offered by companies as they seek to establish dedicated patient bases in the market.
Again, another significant strategy used in Klippel Trenaunay Syndrome Treatment Market is cost leadership. A number of businesses are concentrating on streamlining its operations, engaging suppliers into favorable contracts and setting up effective channels of distribution which will facilitate provision of cost-efficient treatment options for those suffering from KTS. Given that the condition might last long thus making it expensive to manage KTS; most people only prefer cheaply available methods in order to access affordable healthcare services thereby maintaining high market share.
Innovation remains central in KTS Treatment Market where firms spend heavily on research and development with an objective of introducing new therapies as well as technologies. These firms always want to be known as pioneers when it comes to scientific discoveries whether through modern medical devices, directed drug therapies or breakthrough surgical procedures. Therefore, such organizations are seen not only as leaders who meet changing needs among KTS patients through modern solutions but also gain upper hand over others in the industry.
On top of that strategic partnerships and collaborations have been increasingly visible within Klippel Trenaunay Syndrome Treatment Market now days business entities partner with research institutions while health care providers collaborate advocacy groups in order to improve their knowledge base and expand their scope. This leads to increased awareness among a wider range of participants undergoing clinical trials since knowledge transfer has taken place thereby informing decisions regarding overall treatment approaches for better patient outcomes. Hence collaboration strengthens company’s position within the market as well as fosters cooperative environment for all stakeholders within KTS community.
Marketing and branding significantly contribute to market share positioning. As a result, companies have been investing in targeted marketing initiatives to enlighten patients about Klippel Trenaunay Syndrome and the available treatment options. This is important because building a strong brand presence helps create confidence among patients, their family members and medical practitioners. Educational programs, online platforms or participating in relevant meetings help in ensuring effective communication with the target audience hence influencing market shares.
Meanwhile, patient experience holds great importance in KTS Treatment Market as companies embrace customer-centric strategies. It implies offering individualized treatment approaches based on specific needs of Klippel Trenaunay Syndrome patients; delivery of extensive supportive services; prioritizing patient education thereby making the overall wellness and satisfaction of KTS individuals a priority for these firms which can lead to increased loyalty among this group compared to others who may not be satisfied within the same industry.

















Leave a Comment