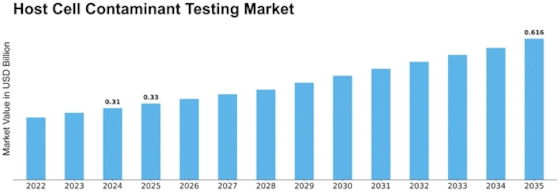
Host Cell Contaminant Testing Size
Host Cell Contaminant Testing Market Growth Projections and Opportunities
The host cell contaminant testing market is experiencing robust growth, propelled by the expanding biopharmaceutical industry. As biopharmaceuticals turn out to be increasingly necessary for medical remedies, the need for rigorous host cell contamination testing is paramount to ensure the protection and efficacy of those superior therapeutics. Stringent regulatory necessities set by the fitness government globally are a major source of pressure for the host cell contamination testing market. Regulatory bodies, along with the FDA and EMA, mandate complete checking out to perceive and quantify Host Cell Contaminants and ensure the purity and safety of biopharmaceutical products. The consciousness of first-rate products inside the biopharmaceutical quarter is a giant issue influencing the host cell contamination testing market. Biopharmaceutical agencies prioritize testing for host cell contaminants to fulfill the very best standards in product fines, stopping detrimental results on sufferers and safeguarding the popularity of their products. Ongoing technological advancements in trying out methods, including polymerase chain reaction (PCR) and mass spectrometry, contribute to the growth of the host cell contaminants testing market. These innovations beautify the sensitivity and specificity of trying out, presenting greater accurate and green identification of Host Cell Contaminants. Host cell contamination testing Biopharmaceutical organizations operating on an international scale are trying to find standardized services to comply with diverse regulatory requirements throughout specific areas. The emphasis on threat mitigation strategies in biopharmaceutical manufacturing is boosting the host cell contamination testing market. Companies prioritize thoroughly trying to perceive and mitigate the dangers associated with Host Cell Contaminants and ensure the reliability and protection of their biopharmaceutical merchandise. Educational initiatives and schooling packages within the biopharmaceutical enterprise play a critical function in the marketplace boom. The awareness of the importance of host cell contamination testing is heightened via educational programs, encouraging biopharmaceutical specialists to adhere to rigorous checking-out protocols. Cost concerns in biopharmaceutical development have an impact on the host cell contamination testing marketplace. Companies are searching for value-effective testing answers that do not compromise on exceptional, leading to a demand for efficient testing offerings that strike a balance between affordability and reliability.




















Leave a Comment