Market Analysis
In-depth Analysis of Distraction Osteogenesis Devices Market Industry Landscape
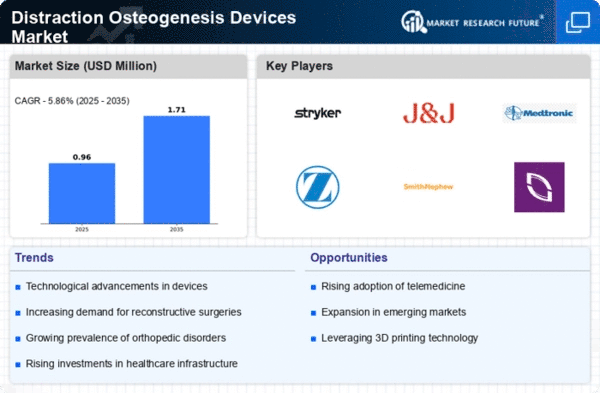
The Distraction Osteogenesis Devices market plays a pivotal role in the orthopedic industry, offering innovative solutions for bone lengthening and deformity correction. The market dynamics are influenced by factors such as advancements in medical technology, increasing prevalence of orthopedic conditions, and a growing demand for minimally invasive surgical techniques. Rapid developments in the healthcare technology are the untiring forces making Disruption Osteogenesis Devices market to grow. Industry is coming out with gadgets that are made to be with high accuracy, ease of operations as well as comfort to patients. Technological advances which are throughput such equipment as robots and computer assistance enhance the high outcome of the distraction osteogenesis operations. Rising down of orthopedic diseases, including the discrepancy of a limb length and congenital deformities which are the main reasons of the market of Distraction Osteogenesis Devices worldwide. Utilization of these technologies is fundamental to the treatment of skeletal deformities requiring bone lengthening or realignment. The orthopedic cases will then be catered for, irrespective of the patients’ age from pediatric to adult. The requirement for intrusive surgical approaches is minimized by the trend of Distraction Osteogenesis Devices towards minimally invasive surgical techniques prevailing. With more and more patients and healthcare workers embracing minimally invasive procedures, which are mainly characterised by smaller cuts, reduced trauma and faster healing rates; the non-invasive surgeries are among the latest methods in medicine. Such distraction osteogenesis tools help in treatment on the basis of osteogenesis technology, which has seen a great level of acceptance by people already. Economic factors in a country scale affect diversity of Distraction Osteogenesis Devices market. The factors as economic stability, currency fluctuation , and the trade policy can influence the production cost and cost of the commodities. Financial constraints could mean issues in medical budgets that might curtail medical providers from getting the best medications and treatment. Ensuring positive patient outcomes and safety is a critical consideration in the Distraction Osteogenesis Devices market. Manufacturers invest in research to develop devices that minimize complications, reduce the risk of infections, and optimize the overall patient experience. Clinical evidence plays a crucial role in establishing the safety and effectiveness of these devices.


















Leave a Comment