Market Analysis
In-depth Analysis of Cone Rod Dystrophy Market Industry Landscape
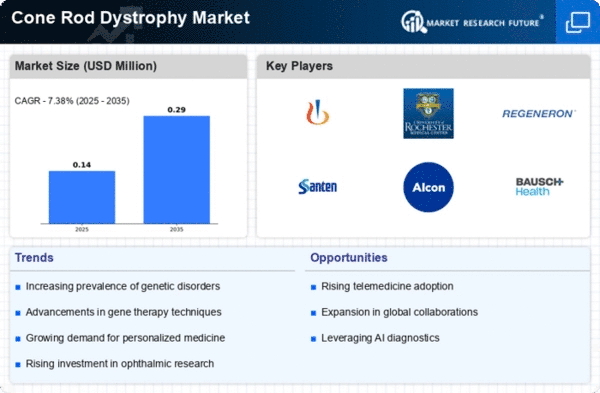
The market for congenital rod dystrophy (CRD) is affected by its rarity. Limited knowledge, diagnostic issues, and a smaller patient population affect CRD medical research, development, and market access. Understanding the genetics of congenital rod dystrophy is a major market driver. hereditary testing and precision medicine have improved diagnosis, allowing for tailored drugs for chronic renal disease (CRD) hereditary anomalies. The CRD market is dynamic due to diagnostic technology advancement. Improved imaging, genetic testing, and biomarker discovery help diagnose congenital rod dystrophy sooner and more accurately, affecting therapy and patient outcomes. Novel drugs and treatment options dramatically impact the CRD market. Research is underway to develop gene therapies, stem cell treatments, and other methods to slow disease progression or restore vision. These initiatives affect competition. CRD market growth comes from patient advocacy groups and increased awareness. Due to increased awareness of chronic renal disease (CRD), healthcare providers and pharmaceutical companies are emphasizing early diagnosis, education, and patient support. Access to expert treatment and healthcare infrastructure quality are crucial to CRD market dynamics. Established ophthalmic institutions with expertise in rare diseases may better meet cataract-related disorder patients' needs. Economic considerations such healthcare spending, insurance coverage, and patient ability to pay affect CRD therapy market dynamics. Novel pharmaceutical costs, reimbursement restrictions, and healthcare spending affect market accessibility and patient outcomes. CRD market dynamics are heavily impacted by regulation. Regulatory clearances, orphan drug designations, and setting standards are key to congenital rod dystrophy medication research, licensing, and marketing. The CRD industry relies on pharmaceutical, academic, and research collaborations. Partnerships help pool resources, share information, and accelerate therapy development. As knowledge grows throughout nations, the CRD market is increasing globally. Global collaborations to promote research, share best practices, and address uncommon disease challenges are shifting market dynamics. Digital and assistive technologies are being used to address chronic respiratory disease (CRD). These innovations will affect the therapeutic environment and market dynamics by making chronic renal disease (CRD) patients' life simpler. When studying market trends, CRD medication pricing and reimbursement methods are crucial. Pharmaceutical companies must balance pricing and accessibility. Reimbursement restrictions affect therapy uptake and market competitiveness.



















Leave a Comment