マイクロサージャリーマーケット 概要
MRFRの分析によると、マイクロサージャリー市場の規模は2024年に26.3億米ドルと推定されています。マイクロサージャリー業界は、2025年に27.82億米ドルから2035年までに48.76億米ドルに成長する見込みで、2025年から2035年の予測期間中に年平均成長率(CAGR)は5.77を示します。
主要な市場動向とハイライト
マイクロサージャリー市場は、技術の進歩と低侵襲手術に対する需要の増加により、 substantial growth の準備が整っています。
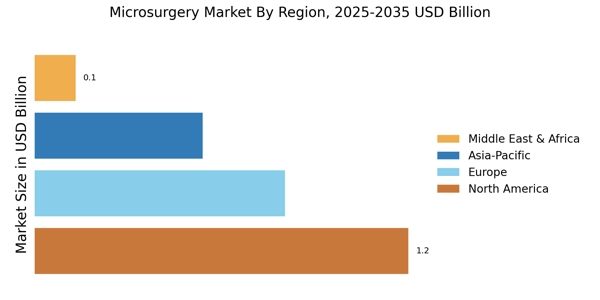
- "北米は、先進的な医療インフラと高い患者意識に支えられ、顕微外科手術の最大の市場であり続けています。
- アジア太平洋地域は、医療投資の増加と人口の増加により、最も成長が早い市場として浮上しています。
- 自由皮弁組織移植が最大のセグメントであり、血管吻合はさまざまな外科手術における応用により急速に成長しています。
- 顕微外科手術における技術革新と外傷ケースの増加は、市場拡大を促進する重要な要因です。"
市場規模と予測
| 2024 Market Size | 2.63 (米ドル十億) |
| 2035 Market Size | 4.876 (米ドル十億) |
| CAGR (2025 - 2035) | 5.77% |
主要なプレーヤー
ストライカー(米国)、メドトロニック(米国)、ジョンソン・エンド・ジョンソン(米国)、B. ブラウン メルスンゲン AG(ドイツ)、カール ストルツ(ドイツ)、オリンパス株式会社(日本)、スミス・アンド・ネフュー(イギリス)、ジンマー・バイオメット(米国)、メディコープ(米国)