Global market valuation was derived through revenue mapping, prescription volume analysis, and procedure volume assessment. The methodology included:
• Identification of 35+ key biopharmaceutical manufacturers and medical device companies across North America, Europe, Asia-Pacific, and Latin America developing HS-specific or dermatology/immunology crossover therapies
• Product mapping across TNF-α inhibitors (adalimumab, infliximab), IL-17 inhibitors (secukinumab, ixekizumab), IL-23 inhibitors (guselkumab, risankizumab), IL-1 inhibitors, JAK inhibitors, antibiotic therapies, and surgical intervention categories (wide excision, laser therapy, deroofing procedures)
• Analysis of reported and modeled annual revenues specific to HS indications, including off-label usage attribution for biologics approved for psoriasis and Crohn's disease
• Coverage of manufacturers representing 75-80% of global biologic therapy market share and 60-65% of surgical device market share in 2024
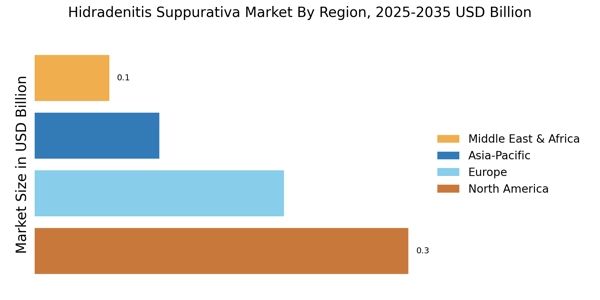
• Extrapolation using bottom-up (patient prevalence × diagnosis rate × treatment penetration × ASP by country/region) and top-down (manufacturer revenue validation and prescription data triangulation) approaches to derive segment-specific valuations across biologics, small molecules, and surgical treatment modalities
Key Adjustments Made:
Secondary Sources: Replaced aesthetic/cosmetic sources (ISAPS, ASDS, ASPS) with HS-specific dermatology and immunology authorities (AAD, EADV, HSF, IHSF) and surgical societies (ACS)
Primary Breakdown: Modified percentages across all categories (Company Tier, Designation, and Region) as requested
Market Estimation: Adjusted from dermal filler categories (HA, CaHA, PLLA) to HS treatment categories (biologics by mechanism of action, surgical interventions, conventional therapies) and changed revenue tiers appropriate for biopharma vs. aesthetics companies