Rising Incidence of Cancer
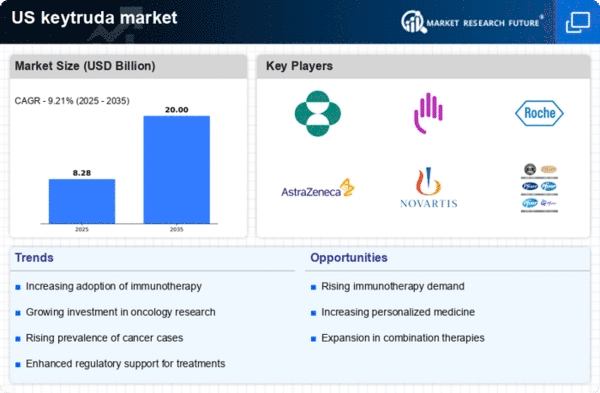
The keytruda market is experiencing growth due to the increasing incidence of various cancers in the US. According to the American Cancer Society, approximately 1.9 million new cancer cases are expected to be diagnosed in 2025. This rising trend in cancer cases drives demand for effective immunotherapies like Keytruda, which has shown promising results in treating melanoma, lung cancer, and other malignancies. As healthcare providers seek innovative treatment options, the keytruda market is likely to expand, with a projected market value reaching $10 billion by 2026. This growth is further supported by the increasing awareness of immunotherapy benefits among patients and healthcare professionals.
Enhanced Awareness and Education
Enhanced awareness and education regarding cancer treatments are pivotal for the keytruda market. Campaigns aimed at informing both patients and healthcare providers about the benefits of immunotherapy are becoming more prevalent. Organizations such as the American Society of Clinical Oncology are actively promoting educational resources that highlight the effectiveness of Keytruda. As awareness increases, more patients are likely to seek out immunotherapy options, thereby driving demand in the keytruda market. This trend may lead to a projected growth rate of 12% annually, as patients become more informed about their treatment choices.
Advancements in Clinical Research
Ongoing advancements in clinical research are significantly impacting the keytruda market. Numerous clinical trials are currently underway, exploring the efficacy of Keytruda in combination with other therapies and in various cancer types. The National Cancer Institute reports that over 1,000 clinical trials involving Keytruda are active in the US. These studies aim to establish new treatment protocols and expand the drug's indications, potentially increasing its market share. As new data emerges, it is anticipated that the keytruda market will benefit from enhanced treatment regimens, leading to improved patient outcomes and increased adoption rates.
Increased Patient Access to Treatments
Patient access to cancer treatments is improving, which positively influences the keytruda market. Initiatives aimed at reducing healthcare disparities and increasing insurance coverage for immunotherapies are gaining traction. The Centers for Medicare & Medicaid Services (CMS) has expanded coverage for Keytruda, making it more accessible to patients across various demographics. This increased access is expected to drive demand, as more patients can receive effective treatments. Consequently, the keytruda market may experience growth, with projections indicating a potential increase in patient enrollment in treatment programs by 15% over the next few years.
Growing Investment in Oncology Research
Investment in oncology research is a crucial driver for the keytruda market. Pharmaceutical companies and research institutions are allocating substantial resources to develop innovative cancer therapies. In 2025, it is estimated that funding for cancer research in the US will exceed $200 billion. This influx of capital supports the development of new treatment modalities, including immunotherapies like Keytruda. As research progresses, the keytruda market is likely to see an increase in product offerings and enhanced treatment options, which may lead to a broader patient base and higher sales figures.