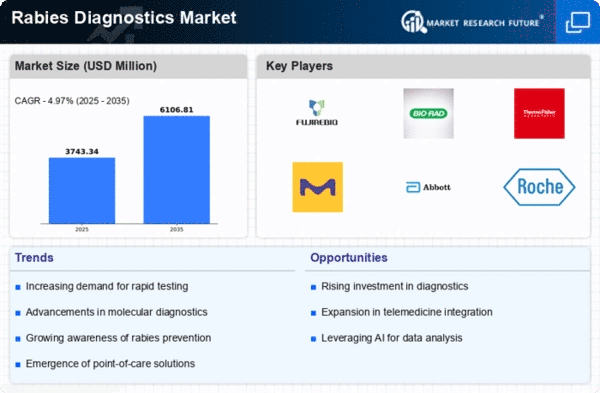
Market Growth Projections
The Global Rabies Diagnostics Market Industry is projected to experience substantial growth in the coming years. With a market value of 2.26 USD Billion in 2024, it is expected to reach 4.15 USD Billion by 2035. This growth reflects a compound annual growth rate of 5.67% from 2025 to 2035. Factors contributing to this upward trajectory include increasing rabies cases, advancements in diagnostic technologies, and enhanced government initiatives. The market's expansion is indicative of a growing recognition of the importance of rabies diagnostics in public health and animal health management.
Global Travel and Trade Expansion
The expansion of global travel and trade is influencing the Global Rabies Diagnostics Market Industry. Increased movement of people and animals across borders raises the risk of rabies transmission, particularly in regions where the disease is endemic. This interconnectedness necessitates robust diagnostic measures to prevent outbreaks. Countries are increasingly recognizing the importance of rabies surveillance and diagnostics as part of their public health strategies. As international travel continues to rise, the demand for effective rabies diagnostics is likely to grow, potentially leading to a compound annual growth rate of 5.67% from 2025 to 2035.
Government Initiatives and Funding
Government initiatives and funding play a crucial role in shaping the Global Rabies Diagnostics Market Industry. Many countries are implementing national rabies control programs, which often include increased funding for diagnostic research and development. For example, the Centers for Disease Control and Prevention in the United States has allocated resources to enhance rabies surveillance and diagnostics. Such initiatives not only promote awareness but also stimulate the market by encouraging the development of innovative diagnostic solutions. As governments prioritize rabies control, the market is likely to experience growth, with projections indicating a market value of 2.26 USD Billion in 2024.
Increasing Incidence of Rabies Cases
The rising incidence of rabies cases globally is a primary driver for the Global Rabies Diagnostics Market Industry. Rabies, a preventable viral disease, continues to pose a significant health threat, particularly in developing regions. According to the World Health Organization, rabies causes approximately 59,000 deaths annually, predominantly in Asia and Africa. This alarming statistic underscores the urgent need for effective diagnostics to facilitate timely treatment and control measures. As awareness of rabies transmission and prevention grows, the demand for diagnostic tools is likely to increase, contributing to the market's expansion.
Technological Advancements in Diagnostic Tools
Technological advancements in diagnostic tools are significantly influencing the Global Rabies Diagnostics Market Industry. Innovations such as rapid diagnostic tests, molecular techniques, and improved serological assays enhance the accuracy and speed of rabies detection. For instance, the development of point-of-care testing devices allows for immediate results, which is crucial in managing rabies exposure. These advancements not only improve diagnostic capabilities but also encourage healthcare providers to adopt new technologies, thereby driving market growth. As these technologies become more accessible, the market is expected to witness substantial growth, particularly in regions with high rabies prevalence.
Rising Pet Ownership and Animal Health Awareness
The increasing trend of pet ownership and heightened awareness of animal health are driving factors in the Global Rabies Diagnostics Market Industry. As more households adopt pets, the risk of rabies transmission from animals to humans rises, necessitating effective diagnostic measures. Public campaigns aimed at educating pet owners about rabies prevention and vaccination further contribute to this trend. This growing awareness is expected to lead to increased demand for rabies diagnostics, as pet owners seek to ensure the health of their animals. Consequently, the market is projected to grow, with an anticipated value of 4.15 USD Billion by 2035.


















Leave a Comment