Market Trends
Key Emerging Trends in the Immunotherapy Drugs market
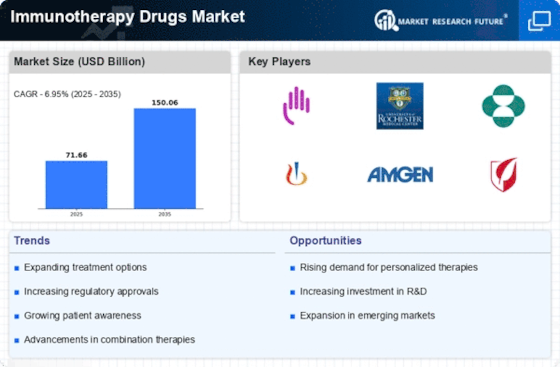
The Immunotherapy Drugs market is witnessing dynamic trends in the field of cancer treatment and autoimmune diseases. As for revolutionary approaches, immunotherapy – the treatment that involves use of ones own immune system to battle diseases has been in a widespread discussion as solution to unmet medical needs. The landscape of the Immunotherapy Drugs market is characterized by several key trends that have emerged in recent years. One of the major trends in Immunotherapy Drugs market is a fast expansion of its uses for cancer treatment. Believe it or not, more and more immunotherapies – immune checkpoint inhibitors, CAR-T cell therapies or cancer vaccines just to name a few become an essential part of treating patients suffering from diverse malignancies. Immunotherapy drugs are crossing the boundaries of oncology and finding their way into other therapeutic areas, such as autoimmune diseases and infectious diseas The inventiveness of immunotherapies in treating a wide range of medical conditions is helping the market diversify and offer more treatment alternatives to patients. Immunotherapy research continues to focus on immune checkpoint inhibitors, including PD-1/PD- L and CTLA -4. Combination therapies and novel checkpoint targets in this category are on-going innovations which provide a positive direction of the Immunotherapy Drugs market improving patient’s response rates. Immunotherapy drugs are being developed and prescribed with the trend towards personalized medicine in mind. Biomarkers, such as PD-L1 expression and tumor mutational burden originate from cells that can be used to identify patients who are likely to respond positively during specific immunotherapies allowing for a more personalized way of treating the patient. A groundbreaking trend in the market for Immunotherapy Drugs is offered by cell and gene therapies, especially C AR-Cell therapy. These therapies consist of the manipulation or altering a patient’s own immune cells so as to target and kill cancerous cell. CAR-T therapies are a game changer in cancer treatment because they have successfully entered the development and commercialisation phase. Another trend to see is in combination therapies involving the use of different types of immunotherapies or when using immunotherapy with traditional treatments such as surgery, chemotherapy or radiology. The rationale of combination approaches is to increase treatment effectiveness, break resistance and deal with the intricately in which immigration system reacts against disease. The availability of biosimilars in the Immunotherapy Drugs market is increasing affordability and accessibility. With patents on some immunotherapy drug drugs expiring, biosimilars provide cost- effective alternatives to potentially increase access of patient’s accessibility into these innovative treatments. One of the trends is the use of immunotherapy in pediatric oncology. Clinical research and trials are concentrating on the safety of immunotherapy drugs in children, as treating pediatric patients can be complicated due to different challenges that arise with these innovative therapies. The Immunotherapy Drugs market is experiencing global expansion as these therapies gain regulatory approvals in various countries. Market players are investing in international collaborations, clinical trials, and regulatory submissions to ensure the accessibility of immunotherapy drugs to patients worldwide, contributing to the globalization of this transformative treatment approach. Patient advocacy groups and increased public awareness are playing a crucial role in shaping the Immunotherapy Drugs market. Patients and their advocates are actively involved in raising awareness, advocating for access to innovative therapies, and participating in clinical trials, influencing the development and adoption of immunotherapy treatments. Despite the remarkable progress, challenges such as managing side effects, understanding resistance mechanisms, and optimizing treatment sequencing remain. However, ongoing research, technological advancements, and a growing understanding of the immune system's intricacies position the Immunotherapy Drugs market for continued evolution and improved patient outcomes.



















Leave a Comment