Market Trends
Key Emerging Trends in the Epigenetics Drugs Diagnostic Technologies Market
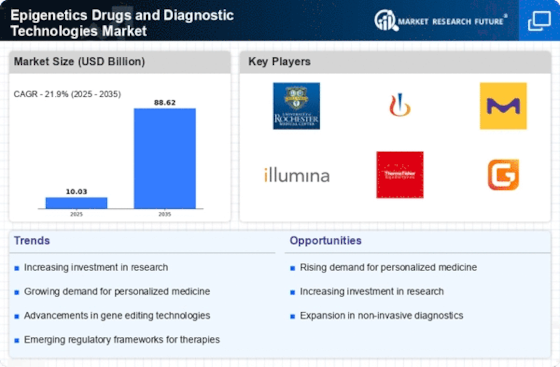
The Epigenetics Drugs Diagnostic Technologies Market is a place where unusual trends are witnessed. These trends reflect a growing recognition of the role of epigenetics in disease development and an increasing demand for innovative diagnostic solutions and treatments. The market consists of drugs and diagnostic technologies that target and analyze epigenetic modifications, which regulate gene expression. Key trends over the past years have characterized this market, reflecting advances in research, better understanding of epigenetic mechanisms, and increased focus on precision medicine.
One hot trend noticeable in the Epigenetics Drugs Diagnostic Technologies Market is the emergence of epigenetic drugs for therapeutic purposes. These drugs are designed to inhibit specific enzymes controlling the modification of chromatin structure with the aim to modify gene expressions patterns and remedy various medical conditions such as cancer, neurological disorders among others. The marketplace has seen a number of Epi-drugs developed and approved thus indicating its potentiality in achieving more targeted treatment approaches that can be effective as well. With continuing improvement in our knowledge about Epigenetics, there is great prospect for new Epi-drugs to be developed thus leading to market growth.
Another trend is the use of diagnostics technologies incorporating epigenetic alterations into integrated testing algorithms or platforms. Thus, epigentic information provides important insights for disease diagnosis, prognosis, and treatment selection. To capture full benefits from personalized medicine concepts DNA-methylation profiling; histone-modification analysis; non-coding RNA expression profiling necessarily should be incorporated into therapeutic design strategies as their absence would deny patients acceptable level care. This movement towards personalized diagnostics based on epigenetic signatures improves accuracy and allows for fine-tuned treatment plans.
Also, they include rising tide of research works aimed at gaining insight into diverse diseases based on their epigenetic foundations taking place within Epigenetics Drugs Diagnostic Technologies Market. Researchers are focusing on finding out if these changes may serve as biomarkers for diagnosis risk assessment or therapy response. This trend is guided by the general switch towards precision medicine in which therapies are developed with the help of individual’s molecular and genetic constitution. The recognition of epigenetic biomarkers offers hope for early intervention that can dramatically change the way diseases are managed.
Moreover, interest in epigenetic editing technologies has increased tremendously within the market, which allows for targeted modifications of specific histone marks. These novel tools enable researchers to study consequences of epigenetic changes on gene expression and explore their therapeutic potential. Still at an early stage, it represents a new frontier in epigenetics field with promise for manipulating disease relevant gene expression as well as treating genetic disorders.
Innovation in Epigenetics Drugs Diagnostic Technologies Market is being driven through partnerships between pharmaceutical companies, diagnostic technology developers and research institutions. These collaborations foster knowledge sharing and resource pooling that accelerate development of novel drugs and diagnostics. This process facilitates translation of scientific discoveries made by different stakeholders from academia to industry leading to improved patient care.
However, there are still challenges facing this industry such as complexity involved in controlling epigenetic processes; designing suitable diagnostic assays and ethical issues related with editing epigenomes. Moreover, there remains a concern about ensuring access to affordable advanced therapeutics based on epigenetics particularly where health systems have limited resources like poor nations.


















Leave a Comment