Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Cardiovascular
Orthopedics
Oncology
Neurology
Hospitals
Ambulatory Surgical Centers
Homecare
Direct Sales
Distributors
Online Sales
North America
Europe
South America
Asia Pacific
Middle East and Africa
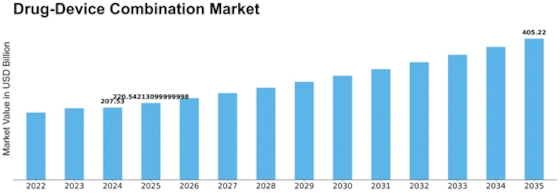
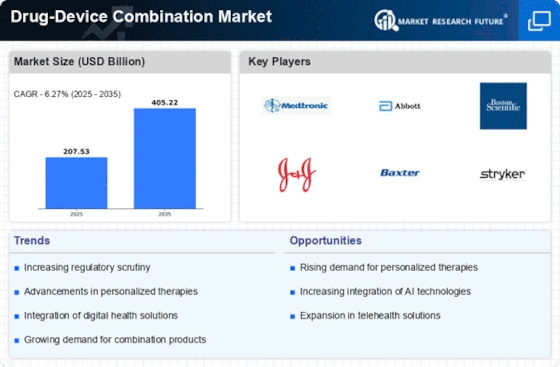
North America Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Drug Device Combination Market by Regional Type
US
Canada
US Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
CANADA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Europe Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Drug Device Combination Market by Regional Type
Germany
UK
France
Russia
Italy
Spain
Rest of Europe
GERMANY Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
UK Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
FRANCE Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
RUSSIA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
ITALY Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
SPAIN Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
REST OF EUROPE Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
APAC Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Drug Device Combination Market by Regional Type
China
India
Japan
South Korea
Malaysia
Thailand
Indonesia
Rest of APAC
CHINA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
INDIA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
JAPAN Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
SOUTH KOREA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
MALAYSIA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
THAILAND Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
INDONESIA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
REST OF APAC Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
South America Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Drug Device Combination Market by Regional Type
Brazil
Mexico
Argentina
Rest of South America
BRAZIL Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
MEXICO Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
ARGENTINA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
REST OF SOUTH AMERICA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
MEA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
Drug Device Combination Market by Regional Type
GCC Countries
South Africa
Rest of MEA
GCC COUNTRIES Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
SOUTH AFRICA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales
REST OF MEA Outlook (USD Billion, 2019-2035)
Drug Device Combination Market by Product Type
Drug-Eluting Stents
Drug-Eluting Balloons
Transdermal Drug Delivery Systems
Inhalation Drug Delivery Devices
Drug Device Combination Market by Application Type
Cardiovascular
Orthopedics
Oncology
Neurology
Drug Device Combination Market by End Use Type
Hospitals
Ambulatory Surgical Centers
Homecare
Drug Device Combination Market by Distribution Channel Type
Direct Sales
Distributors
Online Sales

















Leave a Comment