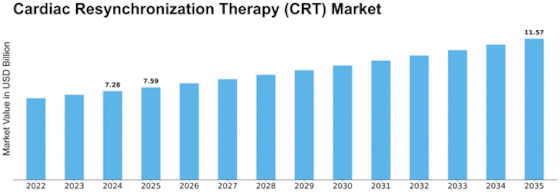
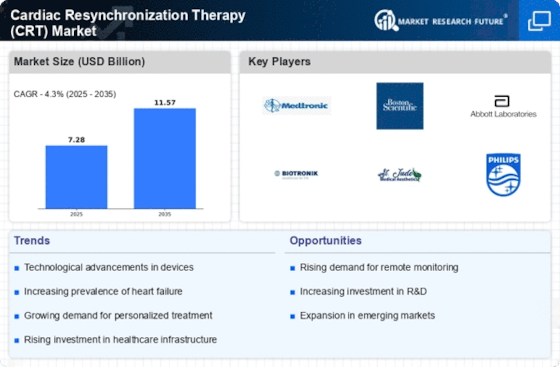
Cardiac Resynchronization Therapy Size
Cardiac Resynchronization Therapy Market Growth Projections and Opportunities
The market for Cardiac Resynchronization Therapy (CRT) is significantly prompted by the growing incidence of coronary heart failure globally. As the prevalence of coronary heart failure continues to boom, the demand for powerful remedy options like CRT has witnessed a parallel surge. The growing older populace, coupled with an upward push in cardiovascular diseases, contributes extensively to the growth of the CRT market. With age being a big risk issue for coronary heart-associated conditions, the growing aged demographic is driving the need for superior cardiac cures. Ongoing technological improvements in CRT devices play a pivotal position in shaping the market. Innovations along with leadless pacing systems, advanced device miniaturization, and improved synchronization algorithms are attracting both sufferers and healthcare companies, fostering market growth. Collaborations and partnerships among key market players make contributions to market growth. These alliances often result in the development of superior and green CRT devices, offering an aggressive area and expanding the overall market. The demand for non-invasive remedies is an extremely good component influencing the CRT market. As sufferers are seeking much less invasive options with decreased recovery times, the demand for CRT devices that offer powerful remedies without good-sized surgical techniques keeps rising. Ongoing research and development (R&D) investments in cardiac therapies, including CRT, fuel market growth. Clinical trials exploring the efficacy and safety of new CRT technologies contribute valuable data, instilling confidence in healthcare professionals and driving adoption. The cost-effectiveness of CRT devices and therapies is crucial for market penetration. Economic factors, including healthcare budget allocations and cost considerations for both providers and patients, play a significant role in determining the market's trajectory. Market dynamics vary across regions due to differences in healthcare infrastructure, economic conditions, and awareness levels. Understanding these geographical variations is essential for stakeholders to tailor their strategies and tap into specific market opportunities. The competitive landscape, characterized by mergers, acquisitions, and market consolidations, shapes the overall CRT market. Key players' strategies influence market trends, pricing, and the availability of innovative products, impacting the demands available to healthcare providers and patients.


















Leave a Comment